Cochlear implants replace the function of damaged sensory hair cells inside the inner ear.
Unlike hearing aids, which mostly make sounds louder, cochlear implants may further improve the clarity of sound and enhance your ability to understand conversations [1,2,3, 14-19].
A cochlear implant system has two parts:
Together, the sound processor and implant bypass the part of the ear that isn’t working, sending sound straight to the hearing nerve.
The Referral Support Tool is a clinical aid for hearing healthcare providers to discuss cochlear implantation as an option to treat hearing loss and promote the referral of suitable patients for further evaluation by specialists at a cochlear implant clinic.
The hearing healthcare provider selects from options that best represent the client such as age range, severity of hearing loss, and four Speech Spatial Qualities (SSQ-49) questionnaire relating to the clarity of sound or how much effort is required for conversation.
The tool uses the personal input and produces an output showing an estimation of a starting binaural aided speech word score test (consonant-nucleus-consonant (CNC) speech discrimination test) and a range of possible binaural hearing performance at one year after implantation. The tool also compares the pre-implantation implantation ratings entered by the user for the four SSQ-49 questions against the proportion of the cochlear implant recipients rating themselves as higher one year post implantation.
The hearing healthcare provider can then use their expertise and this information to counsel the client.
The following links contains information on cochlear implantation candidacy criteria and warnings:
This tool does not recommend or select any specific cochlear implant device. If a particular device is later considered for a patient, clinical use should be consistent with that device’s FDA‑required labeling.
The Referral Support Tool is comprised by several key components:
The Referral Support Tool prompts the hearing healthcare provider to select information that best represents their patient. This information is partially used as an input into the statistical model which estimates the postoperative (one year after implantation) binaural aided CNC speech discrimination score. The following list contains the types of information solicited and the options that the audiologist can select:
The information solicited by the tool remains on the user’s or hearing healthcare provider’s computer and is not transmitted elsewhere or retained. The information inputted is coarse and is not able to be used to link back to the patient.
This tool was developed using best evidence from large high quality clinical datasets. The combined clinical datasets comprise data of approximately 1913 subjects. The constituent datasets are:
The clinical datasets were used in the following ways:
Table 1 records the general demographics of the subjects that were used in the development of the Referral Support Tool.
Table 1 General demographics
| Gender (%) | Age at implantation (years) | ||||||
|---|---|---|---|---|---|---|---|
| Study | #Subject | Female | Male | Unknown | Min | Median | Max |
| Clinical study 1 | 100 | 37.00 | 63.00 | 0.00 | 23 | 71 | 93 |
| Clinical study 2 | 119 | 52.11 | 47.89 | 0.00 | 19 | 68 | 90 |
| Retrospective study | 1282 | 41.88 | 57.95 | 0.17 | 18 | 68 | 102 |
| Registry | 412 | 42.00 | 80.00 | 0.00 | 18 | 59 | 98 |
Severity of hearing loss categories were defined by ranges of unaided air conduction PTA audiometric thresholds (500 Hz, 1000 Hz, 2000 Hz, 4000 Hz). The PTA ranges are:
Subjects of the Retrospective Study were filtered based on their preoperative unaided audiograms conforming to presbycusis style hearing loss or sensorineural hearing loss with high a frequency roll-off. Thereafter they were categorized according to their unaided air conduction PTA. One thousand two hundred and eighty-six subjects remained after filtering and the following data was retrieved for each subject:
Average characteristics of each severity of hearing loss category are calculated by processing the corresponding subject data.The average characteristics of the category are found in Table 2.
Table 2 Characteristics of the severity of hearing loss categories
| Moderate | Severe | Severe to profound | Profound (90 to 100) | Profound (100+) | |
|---|---|---|---|---|---|
| Subjects (#) | 67 | 431 | 296 | 247 | 241 |
| Pure Tone Average (dBHL) | |||||
| PTA Category Lower | 40 | 60 | 80 | 90 | 100 |
| PTA Category Upper | 60 | 80 | 90 | 100 | – |
| PTA Median | 54.25 | 72.5 | 85 | 93.75 | 106.5 |
| Preoperative aided CNC word score (%) | |||||
| CNC Lower | 9.494 | 12 | 8 | 0 | 0 |
| CNC Upper | 69.012 | 44 | 36 | 26 | 12 |
| CNC Median | 40 | 30 | 22 | 12 | 0 |
| Preoperative binaural aided CNC word score (%) | |||||
| CNC Median | 51 | 42 | 35 | 26 | 15 |
| Duration of hearing loss (years) | |||||
| Duration hearing loss | 20 | 23 | 23 | 27 | 29 |
| Duration severe hearing loss | 8.43 | 9.62 | 9.62 | 11.14 | 11.9 |
| Preoperative unaided air conduction audiogram (dBHL) | |||||
| 125 Hz | 36.01 | 37.15 | 39.78 | 50.29 | 67.41 |
| 250 Hz | 34.19 | 40.81 | 46.14 | 58.63 | 78.23 |
| 500 Hz | 34.71 | 46.12 | 54.11 | 67.53 | 87.93 |
| 750 Hz | 37.57 | 53.07 | 63.71 | 76.98 | 96.50 |
| 1000 Hz | 42.86 | 62.80 | 76.68 | 88.69 | 105.07 |
| 1500 Hz | 51.84 | 73.27 | 89.04 | 98.58 | 110.56 |
| 2000 Hz | 60.65 | 89.90 | 99.23 | 106.38 | 114.84 |
| 3000 Hz | 71.23 | 91.57 | 107.15 | 112.27 | 117.90 |
| 4000 Hz | 79.66 | 98.57 | 112.26 | 116.20 | 120.00 |
| 6000 Hz | 87.15 | 104.12 | 114.65 | 118.10 | 120.99 |
| 8000 Hz | 93.72 | 108.22 | 114.31 | 117.97 | 120.88 |
A statistical model estimates the postoperative (one year after implantation) binaural aided CNC score using statistical relationships between preoperative factors and their postoperative score. These relationships are expressed in a linear mathematical equation, which produces a numerical value. An additional algorithm is used to estimate a 90% prediction interval which is a function of the uncertainty or degree of error of the model and the estimated score. The prediction interval provides a range of possible outcomes.
Equations 1 to 3 are the mathematical expression of the model:
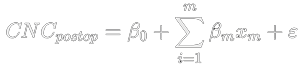 | (1) |
| (2) | |
| (3) |
where CNCpostop is the estimated postoperative CNC word score; β0 is a constant, βm is the coefficient of input variable xm ; ε is the error or uncertainty term; PI is the prediction which is a function of the estimated score and the model uncertainty; and CNCrange is the range of possible outcomes for postoperative CNC word score.
A general linear model (GLM) modelling approach was used to develop the statistical model and estimate the constant and coefficients. More specifically, the Python library Scikit-learn’s Bayesian Ridge Regression algorithm (least squares regression with regularization) was used to estimate the constant and coefficients. Scikit-learn’s Sequential Feature Selector was used to select the input variables that maximize the model’s accuracy. It does this by:
The selected input variables of the model are as follows:
Recipient’s time using the cochlear implant system has been found to be positively correlated with postoperative performance [11]. Holder et al. [12] ran an interventional study and promoted the increase in cochlear implantation system usage for 20 patients. They found that 17 out of the 20 patients increased their average daily usage and the increase in usage was positively correlated with aided hearing performance on CNC words, AzBio sentences and AzBio sentences in noise. While this predictor is a postoperative factor, the use of the model can be used with an assumed average TOA.
The Referral Support Tool use of the model will be with the assumption that the TOA of the hypothetical recipient is 12 hrs/day. This is in line with research conducted by Busch, Vanpoucke and Wieringen [13] that found that the median TOA of adults and seniors is ~ 12 hrs/day.
Subjects of the Clinical Studies were divided into two subsets: training (~66 %) and tuning (~34 %). The model development set was used to identify characteristics that correlate with change in aided CNC scores such as the model input variables. The tuning set was used to identify the best model and to estimate model performance. The performance statistics of the model are recorded in Table 3. These performance statistics can be compared with a naïve model (Table 4) which is the average postoperative (one year after implantation) CNC score being used as the estimate.The model outperforms the naïve model The performance statistics are the coefficient of determination(R2), root mean square error (RMSE) and mean absolute error (MAE).
Table 3 Prototype model accuracy statistics
| Dataset | Samples | R2 | RMSE (%) | MAE (%) | Median (Error) |
|---|---|---|---|---|---|
| Training | 147 | 0.503 | 15.865 | 12.063 | -0.444 |
| Tuning | 72 | 0.531 | 14.691 | 11.765 | -0.688 |
Table 4 Naïve model accuracy statistics
| Dataset | Samples | R2 | RMSE (%) | MAE (%) | Median (Error) |
|---|---|---|---|---|---|
| Training | 147 | – | 22.500 | 18.416 | – |
| Tuning | 72 | – | 21.846 | 17.889 | – |
The SSQ-49 questionnaire measures subjective hearing performance across several dimensions such as directionality of sound, comprehension of speech and quality of sound [20]. Subject data of the Registry (contributing 412 subjects) and Clinical Studies (contributing 200 subjects) were used to substantiate postoperative rankings distributions of SSQ-49 S1Q2, S1Q13, S3Q10 and S3Q15. These postoperative distributions were then used to estimate the proportion of cochlear implant recipients rating themselves as performing better than various preoperative ratings of the same questions. Subjects included were:
Table 5 records the postoperative distributions of the cochlear implant recipients ratings of on the four SSQ-49 questions (S1Q2, S1Q13, S3Q10 and S3Q15).
Table 5 Postoperative SSQ-49 question probability mass distributions
| Notional SSQ Score | S1Q2 (%) | S1Q13 (%) | S3Q10 (%) | S3Q15 (%) |
|---|---|---|---|---|
| 0 | 1.08 | 8.77 | 2.37 | 7.02 |
| 1 | 0.86 | 4.39 | 2.8 | 7.24 |
| 2 | 1.08 | 7.68 | 4.31 | 8.77 |
| 3 | 2.16 | 8.11 | 3.88 | 11.88 |
| 4 | 1.29 | 8.33 | 6.68 | 9.21 |
| 5 | 3.66 | 10.75 | 8.84 | 14.25 |
| 6 | 5.82 | 7.89 | 9.05 | 9.21 |
| 7 | 10.78 | 10.96 | 19.18 | 12.94 |
| 8 | 21.55 | 10.53 | 20.69 | 9.87 |
| 9 | 23.92 | 13.38 | 14.87 | 7.46 |
| 10 | 27.8 | 9.21 | 7.33 | 2.19 |
Comparing to potential or hypothetical preoperative values or ratings on the four SSQ-49 questions, Table 6 records the proportion of cochlear implant recipients rating themselves as performing better.
Table 6 Postoperative SSQ-49 question probability mass distributions
| Proportion of CI users ranking themselves higher (%) | ||||
|---|---|---|---|---|
| Hearing aid patient preoperative rating | S1Q2 (%) | S1Q13 (%) | S3Q10 (%) | S3Q15 (%) |
| 0 | 99.33 | 92.05 | 97.99 | 93.26 |
| 1 | 98.43 | 87.73 | 95.08 | 86.28 |
| 2 | 97.32 | 79.55 | 91.05 | 77.67 |
| 3 | 95.3 | 71.36 | 87.25 | 65.35 |
| 4 | 93.96 | 62.73 | 80.31 | 55.58 |
| 5 | 90.38 | 52.73 | 71.59 | 41.86 |
| 6 | 84.56 | 44.32 | 62.86 | 33.26 |
| 7 | 74.05 | 32.95 | 43.62 | 19.53 |
| 8 | 53.47 | 22.5 | 23.04 | 9.77 |
| 9 | 28.64 | 9.55 | 7.16 | 2.09 |
| 10 | 0 | 0 | 0 | 0 |
Table 7 links the functional outcomes highlighted in the app to academic publications.
| Functional outcome | Ref. |
|---|---|
| Hear alerts and warning sounds |
|
| Watch television and listen to music |
|
| Have a conversation on the phone |
|
| Have a one-on-one conversation |
|
| Index | Document Title | |
|---|---|---|
| [1] | Kumar, R. S., Mawman, D., Sankaran, D., Melling, C., O’Driscoll, M., Freeman, S. M., & Lloyd, S. K. (2016). Cochlear implantation in early deafened, late implanted adults: Do they benefit?. Cochlear implants international, 17(sup1), 22-25. | |
| [2] | Castiglione, A., Benatti, A., Girasoli, L., Caserta, E., Montino, S., Pagliaro, M., … & Martini, A. (2015). Cochlear implantation outcomes in older adults. Hearing, Balance and Communication, 13(2), 86-88. | |
| [3] | Lenarz, M., Sönmez, H., Joseph, G., Büchner, A., & Lenarz, T. (2012). Long-term performance of cochlear implants in postlingually deafened adults. Otolaryngology–Head and Neck Surgery, 147(1), 112-118. | |
| [4] | Severity of Hearing Loss Categories for Binaural Outcomes Prediction Models | |
| [5] | Prototype Binaural English Word Score Outcomes Model | |
| [6] | SSQ Data Analysis for Binaural Referral Support Tool | |
| [7] | Retrospective Study | |
| [8] | Clinical Evaluation of the Cochlear Nucleus CI532 Cochlear Implant in Adults [Internet]. ClinicalTrials.gov identifier: NCT03007472. Bethesda (MD): National Library of Medicine (US). Available from: https://clinicaltrials.gov/study/NCT03007472. Accessed 23 Mar 2026. | |
| [9] | Clinical Investigation Title: Prediction of Outcomes in Adult Cochlear Implant Recipients | |
| [10] | Observation of Benefits for Patients Implanted With a Hearing Implant of the Company Cochlear (IROS) [Internet]. ClinicalTrials.gov identifier: NCT02004353. Bethesda (MD): National Library of Medicine (US). Available from: https://clinicaltrials.gov/study/NCT02004353. Accessed 23 Mar 2026. | |
| [11] | Holder, J. T., Dwyer, N. C., & Gifford, R. H. (2020). Duration of processor use per day is significantly correlated with speech recognition abilities in adults with cochlear implants. Otology & neurotology: official publication of the American Otological Society, American Neurotology Society [and] European Academy of Otology and Neurotology, 41(2), e227. | |
| [12] | Holder, J. T., & Gifford, R. H. (2021). Effect of increased daily cochlear implant use on auditory perception in adults. Journal of Speech, Language, and Hearing Research, 64(10), 4044-4055. | |
| [13] | Busch, T., Vanpoucke, F., & van Wieringen, A. (2017). Auditory environment across the life span of cochlear implant users: Insights from data logging. Journal of Speech, Language, and Hearing Research, 60(5), 1362-1377. | |
| [14] | Bento, R. F., Brito Neto, R., Castilho, A. M., Gómez, V. G., Giorgi, S. B., & Guedes, M. C. (2004). Auditory results with multichannel cochlear implant in patients submitted to cochlear implant surgery at Medical School, Hospital das Clínicas, University of Sao Paulo. Braz J Otorhinolaryngol, 70, 632-7. | |
| [15] | Zhao, F., Stephens, S. D. G., Sim, S. W., & Meredith, R. (1997). The use of qualitative questionnaires in patients having and being considered for cochlear implants. Clinical Otolaryngology & Allied Sciences, 22(3), 254-259. | |
| [16] | Wick, C. C., Kallogjeri, D., McJunkin, J. L., Durakovic, N., Holden, L. K., Herzog, J. A., … & CI532 Study Group. (2020). Hearing and quality-of-life outcomes after cochlear implantation in adult hearing aid users 65 years or older: a secondary analysis of a nonrandomized clinical trial. JAMA Otolaryngology–Head & Neck Surgery, 146(10), 925-932. | |
| [17] | Porps, S. L., Bennett, D. M., Gilden, J., Ravelo, K., Buck, B., Reinhart, P., & Hong, R. S. (2023). Effects of an evidence-based model for cochlear implant aftercare delivery on clinical efficiency and patient outcomes. Cochlear Implants International, 1-10. | |
| [18] | Reed, C. M., & Delhorne, L. A. (2005). Reception of environmental sounds through cochlear implants. Ear and Hearing, 26(1), 48-61. | |
| [19] | Di Nardo, W., Anzivino, R., Giannantonio, S., Schinaia, L., & Paludetti, G. (2014). The effects of cochlear implantation on quality of life in the elderly. European Archives of Oto-Rhino-Laryngology, 271, 65-73. | |
| [20] | Gatehouse, S., & Noble, W. (2004). The speech, spatial and qualities of hearing scale (SSQ). International Journal of Audiology, 43(2), 85-99. | |
Shukla A, et al. 2020, Otolaryngology–head and neck surgery; 162(5):622-33.
Methodology
Systematic literature review to summarize the current state of the literature exploring the association between hearing loss and social isolation and/or loneliness.
Findings and Recommendations
Bott A, et al. 2021, International Journal of Audiology; 60:30-46.
Methodology
Scoping review to map and evaluate the extent, range, and nature of research examining the relationship between hearing loss, social isolation, and loneliness in adults across the lifespan
Findings and recommendations
Golub, J. S. et.al.(2019). Association of Audiometric Age-Related Hearing Loss With Depressive Symptoms Among Hispanic Individuals. JAMA otolaryngology– head & neck surgery, 145(2), 132–139
Methodology
Cross-sectional study on 5,328 Hispanic adults to investigate whether objectively measured age-related hearing loss (HL) is associated with clinically significant depressive symptoms
Findings and recommendations
Nachtegaal J, Smit JH, Smits C, et al. The association between hearing status and psychosocial health before the age of 70 years: results from an internet-based national survey on hearing. Ear Hear. 2009;30(3):302-312.
Methodology
Cross sectional cohort study of 1,511 patients
Findings and recommendations
Greenberg PE, et al. Pharmacoeconomics. 2021;39(6):653-665
Methodology
Combined original analyses and literature based estimates based on the national suvey of drug use and the Optum health administrative claim database to update costs of Major Depressive Disorder (MDD) for adults in USA for 2018
Findings and recommendations
Bigelow RT, et al. JAMA Netw Open. 2020;3(7)e2010986.
Methodology
Cross sectional study on adults in USA using data from 2017 National Health Interview Survey, 25 665 adults included in analysis to investigate the association between HL, psychological distress, and mental health care utilization among adults in the United States.
Findings and recommendations
Huber M, et al. Ear Hear. 2025; Published online September 15, 2025.
Methodology
Cohort study in two tertiary research centers of 61 adult patients indicated for CI in Austria and Germany to investigate whether an improvement in hearing after cochlear implantation (CI) was associated with changes in depression and cognitive performance
Findings and recommendations
McIlhiney P, et al. Clin Otolaryngol. 2025;50(5):848-855.
Methodology
Controlled, observational study of 87 adults in three groups, 44 normal hearing, 26 fitted with CI and 17 with untreated hearing loss in Australia to examine how cochlear implantation affects depression, anxiety and stress levels.
Findings and recommendations
Gerst-Emerson K, et al. Loneliness as a public health issue: the impact of loneliness on health care utilization among older adults. Am J Public Health. 2015;105(5):1013–1019.
Methodology
Health and Retirement Study (HRS) of 3,530 US community-dwelling adults aged 60+ to examine whether loneliness is associated with increased health care utilization
Findings and recommendations
Sterling MR, Lin FR, Jannat-Khah DP, Goman AM, Echeverria SE, Safford MM. Hearing loss among older adults with heart failure in the United States: data from the National Health and Nutrition Examination Survey. JAMA Otolaryngol Head Neck Surg. 2018;144(3):273–275
Methodology
Cross sectional analysis on US adults over 70
Findings and recommendations
Hull RH, Kerschen SR. The influence of cardiovascular health on peripheral and central auditory function in adults: a research review. American Journal of Audiology. 2010 Jun;19(1):9-16
Methodology
Comparative research review
Findings and recommendations
Papadopoulou AM, Papouliakos S, Karkos P, Chaidas K. The impact of cardiovascular risk factors on the incidence, severity, and prognosis of sudden sensorineural hearing loss (SSHL): a systematic review. Cureus. 2024;16(4):e58377
Methodology
Systematic literature review of 24 studies with 61,060 patients
Findings and recommendations
Wang C, Xu F, Chen M, Chen X, Li C, Sun X, Zhang Y, et al. Association of obstructive sleep apnea-hypopnea syndrome with hearing loss: a systematic review and meta-analysis. Front Neurol. 2022;13:1017982
Methodology
Systematic review and meta-analysis of 10 studies with 7,867 participants
Findings and recommendations
Jo YS, Lee JM. Assessing the impact of apnea duration on the relationship between obstructive sleep apnea and hearing loss. PLoS One. 2024;19(12):e0315580
Methodology
Retrospective chart review of 90 patients
Findings and recommendations
Kayabasi, S., Hizli, O. & Yildirim, G. The association between obstructive sleep apnea and hearing loss: a cross-sectional analysis. Eur Arch Otorhinolaryngol 276, 2215–2221 (2019)
Methodology
Systematic review and meta-analysis of 12 studies with 9,079 participants
Findings and recommendations
Gopinath B, Flood VM, Teber E, McMahon CM, Mitchell P. Dietary intake of cholesterol is positively associated, and use of cholesterol-lowering medication is negatively associated with prevalent age-related hearing loss. J Nutr. 2011;141(7):1355–1361
Methodology
Population based cohort study of 2,447 adults
Findings and recommendations
Silky S, Singh V, Gupta DK, Chaudhary AK, Yadav R, Kumar R, Siva S, Jain RK. A study of relationship between serum lipids and sensorineural hearing loss. Indian J Otolaryngol Head Neck Surg. 2023;75(Suppl 2):S578–S583.
Methodology
Cross sectional observational study of 150 patients aged 15-60 with hearing loss
Findings and recommendations
Yu Y, Yang ZC, Wang LX. Triglyceride to high-density lipoprotein cholesterol ratio and sensorineural hearing loss in community-dwelling adults: an NHANES analysis. Yonsei Med J. 2024;65(12):741–751.
Methodology
Cross sectional analysis of 1,148 US aduts
Findings and recommendations
Ramatsoma H, Patrick SM. Hypertension associated with hearing loss and tinnitus among hypertensive adults at a tertiary hospital in South Africa. Front Neurol. 2022;13:857600
Methodology
Quantitative cross-sectional study of 106 hypertensive adults and 92 age matched controls
Findings and recommendations
Toyama K, Mogi M. Hypertension and the development of hearing loss. Hypertens Res. 2022;45:172–174
Methodology
Commentary to study by Miyata et al.
Findings and recommendations
Hou Y, Liu B. Relationship Between Hypertension and Hearing Loss: Analysis of the Related Factors. Clin Interv Aging. 2024;19:845–856.
Methodology
Observational study of 517 adult patients with and without hypertension
Findings and recommendations
Agmon M, Lavie L, Doumas M. The Association between Hearing Loss, Postural Control, and Mobility in Older Adults: A Systematic Review. Journal of the American Academy of Audiology. 2017;28(6):575-88.
Methodology
Systematic literature review to systematically review the evidence on the relationship between hearing loss (HL) and postural control/mobility in older adults
Findings and recommendations
Foster JL et al., Trends Hear. 2022;26:23312165221144155.
Methodology
Systematic review and meta-analysis on adults aged ≥60 years with hearing loss and objective measures of postural stability or gait. 27,847 across 25 studies in multiple countries
Findings and recommendations
Haddad YK, et al., Inj Prev. 2024; 30(4):272-276.
Methodology
Observational study using estimated health expenditures on 9,199 patients in USA
Findings and recommendations
Goman AM et al., Lancet Public Health. 2025;10(6):e492-e502
Methodology
Secondary analysis of the ACHIEVE randomized controlled trial on 977 adults aged 70-84 in USA
Findings and recommendations
Louza J, et al. Audiol Neurotol. 2019;24(5):245-252
Methodology
Prospective clinical trial on 33 adults in Germany with CI assessing postural control.
Findings and recommendations
Lin FR, Metter EJ, O’Brien RJ, Resnick SM, Zonderman AB, Ferrucci L. Hearing loss and incident dementia. Arch Neurol. 2011;68(2):214-220. doi:10.1001/archneurol.2010.362
Methodology
Prospective cohort study of 639 dementia-free US participants aged 36–90 to investigate whether hearing loss is prospectively associated with the development of all-cause dementia and Alzheimer’s disease (AD) in older adults.
Findings and recommendations
Yu RC, Proctor D, Soni J, et al. Adult-onset hearing loss and incident cognitive impairment and dementia – A systematic review and meta-analysis of cohort studies. Ageing Res Rev. 2024;98:102346
Methodology
Systematic review and meta-analysis to comprehensively evaluate whether adult-onset hearing loss is associated with increased risk of cognitive impairment and dementia
Findings and recommendations
Livingston G, Huntley J, Liu KY, et al. Dementia prevention, intervention, and care: 2024 report of the Lancet standing Commission. Lancet. 2024;404(10452):572-628.
Methodology
Synthesize findings from systematic reviews, meta-analyses, cohort studies, and RCT’s to update the 2020 report with new evidence on dementia prevention
Findings and recommendations
Wimo A, Seeher K, Cataldi R, et al. The worldwide costs of dementia in 2019. Alzheimers Dement. 2023;19(7):2865-2873. doi:10.1002/alz.12901
Methodology
Leverage prevalence data from IHME and UN population datasets to estimate the global societal costs of dementia in 2019
Findings and recommendations
Mukadam N, Anderson R, Knapp M, et al. Effective interventions for potentially modifiable risk factors for late-onset dementia: a costs and cost-effectiveness modelling study. Lancet Healthy Longev. 2020;1(1):e13-e20.
Methodology
Cost modelling study to evaluate the cost-effectiveness of interventions targeting nine modifiable risk factors for late-onset dementia
Findings and recommendations
Seo HW, Ryu S, Han SY, Lee SH, Chung JH. Cochlear Implantation Is Associated With Reduced Incidence of Dementia in Severe Hearing Loss. Ear Hear. 2025;46(5):1189-1196.
Methodology
Observational population study on 52,219 South Korean adults with severe hearing loss and 1,280,788 individuals with normal hearing to investigate whether auditory rehabilitation—specifically cochlear implants (CI) and hearing aids (HA)—reduces the long-term risk of dementia
Findings and recommendations
Huddle, M. G., Goman, A. M., Kernizan, F. C., Foley, D. M., Price, C., Frick, K. D., & Lin, F. R. (2017). The Economic Impact of Adult Hearing Loss: A Systematic Review. JAMA Otolaryngology–Head & Neck Surgery, 143(10), 1040–1048
Methodology
Systematic literature review to systematically review and summarize existing data on the direct and indirect economic costs associated with adult hearing impairment
Findings and recommendations
Mohr, P. E., Feldman, J. J., Dunbar, J. L., McConkey-Robbins, A., Niparko, J. K., Rittenhouse, R. K., & Skinner, M. W. (2000). The Societal Costs of Severe to Profound Hearing Loss in the United States. International Journal of Technology Assessment in Health Care, 16(4), 1120–1135.
Methods
Incidence-based cohort-survival model using a Markov process to provide a comprehensive, national estimate of the lifetime economic burden of severe to profound hearing impairment (HI) in the U.S
Findings and recommendations
Shukla A, et al. 2020, Otolaryngology–head and neck surgery; 162(5):622-33.
Methodology
Systematic literature review to summarize the current state of the literature exploring the association between hearing loss and social isolation and/or loneliness.
Findings and Recommendations
Bott A, et al. 2021, International Journal of Audiology; 60:30-46.
Methodology
Scoping review to map and evaluate the extent, range, and nature of research examining the relationship between hearing loss, social isolation, and loneliness in adults across the lifespan.
Findings and recommendations
Golub, J. S. et.al.(2019). Association of Audiometric Age-Related Hearing Loss With Depressive Symptoms Among Hispanic Individuals. JAMA otolaryngology– head & neck surgery, 145(2), 132–139
Methodology
Cross-sectional study on 5,328 Hispanic adults to investigate whether objectively measured age-related hearing loss (HL) is associated with clinically significant social isolation symptoms in older Hispanic adults.
Findings and recommendations
Nachtegaal J, Smit JH, Smits C, et al. The association between hearing status and psychosocial health before the age of 70 years: results from an internet-based national survey on hearing. Ear Hear. 2009;30(3):302-312.
Methodology
Cross-sectional study on 925 working Dutch adults to investigate the relationship between hearing status and need for recovery after work and whether psychosocial work characteristics (job demands and job control) influence this relationship.
Findings and recommendations
Ray J, et al. Association of cognition and age-related hearing impairment in the English Longitudinal Study of Ageing. JAMA Otolaryngol Head Neck Surg. 2018;144(10): 876–882.
Methodology
Cross-sectional analysis on 7,385 English adults to examine the link between age-related hearing impairment (ARHI) and cognitive decline in older adults in the UK
Findings and recommendations
Gerst-Emerson K, et al. Loneliness as a public health issue: the impact of loneliness on health care utilization among older adults. Am J Public Health. 2015;105(5):1013–1019.
Methodology
Health and Retirement Study (HRS) of 3,530 US community-dwelling adults aged 60+ to examine whether loneliness is associated with increased health care utilization
Findings and recommendations
Flowers L, Shaw J, Farid M. Medicare spends more on socially isolated older adults. Insight on the Issues. Nov 2017.
Methodology
Health and Retirement Study (HRS) of 5,270 US community-dwelling adults aged 65+ to investigate whether social isolation among older adults is associated with increased Medicare spending.
Findings and recommendations
Hori K, Shah R, Paladugu A, et al. Social Outcomes Among Adults With Hearing Aids and Cochlear Implants: A Systematic Review and Meta-Analysis. JAMA Otolaryngol Head Neck Surg. 2025;151(8):806-816. doi:10.1001/jamaoto.2025.1777
Methodology
Systematic literature review and meta-analysis to evaluate the impact of hearing rehabilitation devices
Findings and recommendations
Bainbridge KE, Cowie CC, Gonzalez F 2nd, Hoffman HJ, Dinces E, Stamler J, Cruickshanks KJ. Risk factors for hearing impairment among adults with diabetes: The Hispanic Community Health Study/Study of Latinos (HCHS/SOL). J Clin Transl Endocrinol. 2016;6:15–22.
Methodology
Cross-sectional analysis of 3,384 adults with diabetes
Findings and recommendations
Caballero-Borrego M, Andujar-Lara I. Type 2 diabetes mellitus and hearing loss: A PRISMA systematic review and meta-analysis. Otolaryngol Head Neck Surg. 2025;173(5):1041–1053
Methodology
Systematic review and meta-analysis of 17 papers
Findings and recommendations
Deng Y, Chen S, Hu J. Diabetes mellitus and hearing loss. Mol Med. 2023;29(1):141.
Methodology
Narrative review
Findings and recommendations
Villarroel, M. A. B. D., Blackwell, D. L., & Jen, A. (2019). Tables of summary health statistics for U.S. adults: 2018 National Health Interview Survey. National Center for Health Statistics
Methodology:
Health statistics from US
Findings and Recommendations
Simpson AN, Matthews LJ, Cassarly C, Dubno JR. Time From Hearing Aid Candidacy to Hearing Aid Adoption: A Longitudinal Cohort Study. Ear Hear. 2019 May/Jun;40(3):468-476.
Objective:
Assess time from hearing aid candidacy to adoption
Methodology:
Longitudinal study of 1.530 adults in US
Findings and Recommendations
ASHA audiology information series, comorbidities and hearing loss. ASHA 2021.
Objective:
Summarise risk factors and health impact from hearing loss
Methodology:
Informational review
Findings and Recommendations
Reed NS, Altan A, Deal JA, et al. Trends in Health Care Costs and Utilization Associated With Untreated Hearing Loss Over 10 Years. JAMA Otolaryngol Head Neck Surg. 2019;145(1):27–34.
Objective:
Evaluate if untreated hearing loss associated with higher health care costs and utilization
Methodology:
Retrospective study of insurance database of 154.414 subjects
Findings and Recommendations
Rein DB et al. The prevalence of bilateral hearing loss in the United States in 2019: a small area estimation modelling approach for obtaining national, state, and county level estimates by demographic subgroup. The Lancet regional health, v30, 2024.
Objective:
Re-estimate rates of bilateral hearing loss Nationally, and create new estimates of hearing loss prevalence at the U.S. State and County levels
Methodology:
Small Area Estimation Modelling
Findings and Recommendations